
By Xam Riche on April 29, 2026 • 13 min read
This article is for informational and educational purposes only and does not constitute medical advice. Always consult with a qualified healthcare professional before using mechanism-based gut or pain information to make diagnosis or treatment decisions.
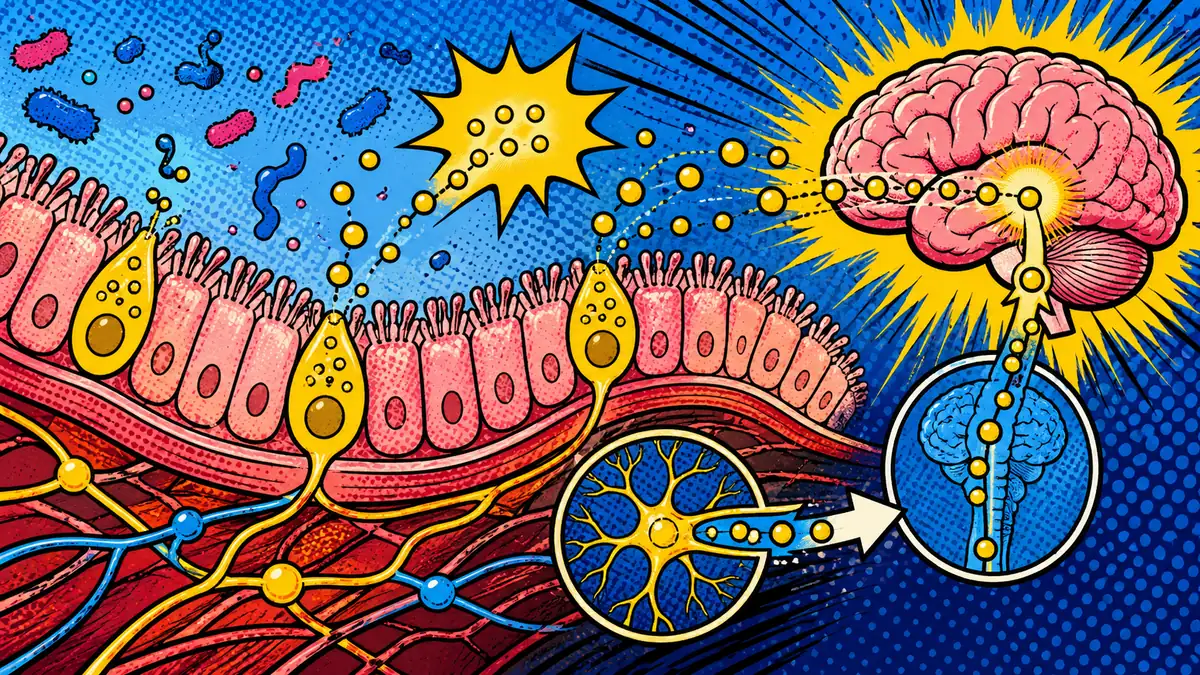
If serotonin and IBS-D explains one part of your symptoms and visceral pain amplification explains another part, the missing question is often smaller: what sits in between? One answer is a set of specialized gut sensory cells called enterochromaffin cells. They help translate stretch, nutrients, microbes, and local irritation into serotonin-related signaling that can change how the gut moves and how loudly the gut is felt.
Short answer: enterochromaffin cells are specialized serotonin-producing gut cells that help connect luminal or microbial information with neural and local signaling pathways. That matters because serotonin in the gut helps influence motility, secretion, and sensation, which is one reason the same broad signaling lane can show up as urgency-heavy IBS-D in one reader and pain-amplification or visceral hypersensitivity in another 1 2.
This page is for you if you already know the bigger gut-brain story but still want the missing cell-level bridge between gut inputs, serotonin, urgency, and pain.
Use a different page first if you mainly need the broad signaling map, a microbiota explainer, or a narrow IBS-D treatment route. Start with gut-brain signaling and appetite, microbiota-gut-brain axis explained, or serotonin and IBS-D.
Health note: this page explains one mechanism lane. It is not a diagnosis and it is not proof that one serotonin, microbiome, or supplement theory is the whole answer.
Use this selector before staying in cell-level mechanism mode.
| If the loudest issue is... | Best next read | Why |
|---|---|---|
| Fear, anticipation, low mood, or the symptom loop is taking over daily life | IBS, anxiety, and depression | It owns the mood-loop and care-lane conversation without saying symptoms are imaginary. |
| Urgency, diarrhea, fast transit, or serotonin-targeted IBS-D options | Serotonin and IBS-D | It keeps urgency-heavy patterns in the IBS-D mechanism and treatment lane. |
| How gut contents become serotonin, motility, urgency, and pain signals | This page | It owns the upstream EC-cell mechanism bridge. |
| Pain volume, stress sensitivity, sex or hormone context, or persistent visceral pain | Stress, sex, and chronic visceral pain | It routes pain amplification into the pain-threshold anchor. |
| The pattern is mixed, persistent, medication-adjacent, or hard to summarize | Doctor visit prep for IBS next steps | It turns mechanism reading into a cleaner clinician handoff. |
The current shelf already has two strong downstream pages:
What many readers still do not have is the cell-level bridge between those two routes.
That bridge matters because gut-brain articles can otherwise flatten the story into vague language:
Enterochromaffin cells help keep the explanation grounded. They are one of the places where gut contents become interpretable signals. They do not explain everything, but they explain why bowel speed, secretion, urgency, and pain can sit in the same neighborhood without being the same symptom.
If you are starting higher up the ladder and still need the broader map first, the parent bridge is gut-brain signaling and appetite. If you want the upstream microbial context, go to microbiota-gut-brain axis explained.
Enterochromaffin cells show up in IBS conversations because they sit near a busy conversion point: gut contents on one side, gut nerves and local immune signals on the other.
That does not mean EC cells "cause IBS" by themselves. It means they help explain why several IBS-style outputs can share part of the same signaling neighborhood:
| Downstream output | Why EC-cell signaling can be relevant | Better next route |
|---|---|---|
| Urgency and fast transit | Gut serotonin can influence motility and secretion, which can make the mechanism relevant to IBS-D urgency. | Serotonin and IBS-D |
| Pain that feels louder than the visible gut event | Serotonin-related sensory signaling can sit upstream of visceral hypersensitivity and pain amplification. | Visceral hypersensitivity in IBS |
| Stress-sensitive or context-sensitive pain | EC-cell signaling is one lane inside a larger stress, immune, microbiota, and gut-brain system. | Stress, sex, and chronic visceral pain |
| Microbiome and metabolite questions | EC cells can respond to microbial and luminal cues, but they are not the whole microbiota-gut-brain axis. | Microbiota-gut-brain axis explained |
The reader-safe version is this: EC cells help make the mechanism more concrete, but the symptom route still depends on whether urgency, pain, microbiome context, or red flags are the main problem.
Enterochromaffin cells, often shortened to EC cells, are specialized enteroendocrine cells scattered through the gut lining. Reviews describe them as major serotonin-producing sensory cells that detect cues from the gut lumen and coordinate communication across the gut wall 3 4.
That sounds abstract until you translate the job:
This is why they belong in a gut-brain explanation rather than in an obscure cell-biology footnote. They sit at a useful conversion point between the gut environment and the nervous system's interpretation of that environment.
One reason the field gets confusing is that readers encounter overlapping terms such as enteroendocrine cells, neuropod cells, or gut sensory cells in headlines. The cleanest editorial move is not to force those words to mean the same thing. It is to say that EC cells are one serotonin-centered part of that sensory gut interface, and that this article is about that narrower lane.
The gut is not waiting passively for damage before it sends information. Enterochromaffin cells help interpret ordinary gut events such as stretch, nutrient arrival, microbial products, and local inflammatory context 5 6.
Once activated, EC-cell signaling can affect several routes at once:
That matters because a gut symptom is not only "food went in, symptom came out." There is a translation layer in between. EC cells are one part of that translation layer.
The point of this diagram is not to say EC cells control the whole gut-brain axis. It is to show why a cell-level lane can have more than one output. The same upstream signaling story can feed a bowel-speed problem in one person and a pain-volume problem in another.
Serotonin in the gut is not only about one feeling or one diagnosis. Reviews on gastrointestinal serotonin describe it as relevant to motility, secretion, sensation, and inflammatory signaling 7. That is why serotonin can be useful in both diarrhea-heavy and pain-heavy conversations without meaning the two conversations are identical.
If the dominant pattern is:
then the more useful downstream page is serotonin and IBS-D. That page explains why clinicians may discuss 5-HT3-related IBS-D treatment lanes and how to compare them against other IBS-D options.
If the dominant pattern is:
then the better downstream page is stress, sex, and chronic visceral pain. That page explains why gut sensation can become amplified without implying the pain is imaginary.
The shared EC-cell lane helps explain why these patterns can overlap. The route still depends on which output is loudest.
This page is upstream of the IBS-D article.
Serotonin and IBS-D asks a practical downstream question: when urgency, loose stool, and fast transit dominate, how should a reader think about serotonin-linked IBS-D treatment lanes under clinician guidance?
This page asks a mechanism question: how do gut sensory cells help translate stretch, luminal contents, microbial cues, and local context into signals that can affect motility, secretion, urgency, and pain?
That difference keeps the boundaries clean:
So if your main question is "What should I discuss for IBS-D urgency?", go to serotonin and IBS-D. If your main question is "Why can one cell-level lane touch both urgency and pain?", stay here.
This is where the article needs restraint.
The EC-cell hyperalgesia review and related IBS review literature connect EC cells, serotonin release, microbial or inflammatory cues, and pain-related signaling 8 9. That is important, but it is not the same as saying EC cells have become a routine stand-alone clinical test for IBS or chronic abdominal pain.
The useful translation is:
This is one reason readers should not overread mechanism articles. A mechanistic bridge can be accurate and still not tell you the one best therapy by itself. If you need the broader treatment ladder after understanding the mechanism, go to IBS treatment.
This page explains several useful things well.
That boundary matters because gut-brain writing goes wrong when mechanism gets promoted into certainty. The better use of mechanism is better routing.
If food-trigger work is still messy, the next route may be when low FODMAP does not work: next steps, not more cell biology. If the symptom pattern already looks like a broader microbiota question, go back up to microbiota-gut-brain axis explained.
This article should narrow the next question, not widen it forever.

| Mechanism or symptom question | Best next read | Why |
|---|---|---|
| The broad gut-brain system is still unclear | Microbiota-gut-brain axis explained | Better for neural, immune, serotonin, microbial metabolite, and stress lanes together. |
| Bowel urgency and fast transit are loudest | Serotonin and IBS-D | Better for urgency-heavy IBS-D routing and clinician-guided treatment-lane comparison. |
| Urgency mainly happens after meals | Urgency after meals | Better for sorting caffeine, fat load, sugar alcohols, meal timing, IBS-D, and red flags before mechanism speculation. |
| Normal digestion feels painful | Visceral hypersensitivity in IBS | Better first if the main question is why ordinary gut signals can feel painful. |
| Pain is stress-sensitive, hormone-context sensitive, or persistent | Stress, sex, and chronic visceral pain | Better for pain amplification inside a wider gut-brain and life-context frame. |
| You need clinician-guided escalation rather than more mechanism | IBS treatment | Better for comparing diet, medication, gut-brain, and pain-focused options. |
| Your notes are mixed, persistent, medication-adjacent, or hard to summarize | Doctor visit prep for IBS next steps | Better for turning mechanism questions into a clear clinician handoff. |
Notice what is missing from the table: a supplement shortcut. This mechanism is useful because it helps route the next question, not because it proves a consumer-level EC-cell treatment plan.
Download: Gut Pain Signaling Discussion Guide Use this one-page guide if you want a cleaner conversation about whether the next route is urgency-heavy IBS-D support, visceral-pain support, or a mixed pattern discussion.
Download: EC Cell, Serotonin, and Pain Router Use this quick router if you want the best next page after the mechanism makes sense.
Mechanism reading should never delay evaluation of red flags. Do not keep routing yourself through serotonin, microbiota, or stress explanations if you have:
Use mechanism pages to ask sharper questions, not to explain away danger signs.
Enterochromaffin cells are one of the gut's useful translation layers. They help convert luminal, microbial, and mechanical gut events into serotonin-related signaling that can influence motility, secretion, and sensation 10 11.
That helps explain why urgency-heavy IBS-D and visceral pain amplification can share some upstream biology without being the same problem. The mechanism is real. It is also not the whole story.
The practical move is pattern matching:
Xam Riche is a gut health solopreneur and founder of YourFitNature, dedicated to helping people navigate digestive wellness through evidence-based information and personal experience. After years of struggling with IBS and bloating, Xam discovered the transformative power of the low FODMAP diet and now shares practical, science-backed guidance to help others find relief. While not a medical professional, Xam combines extensive research with lived experience to create accessible, empowering resources for the gut health community. Learn more about our mission
As an affiliate, we may earn from qualifying purchases.
Showing 10 of 127